News and
Announcements
Stay up to date on all our news and market announcements.
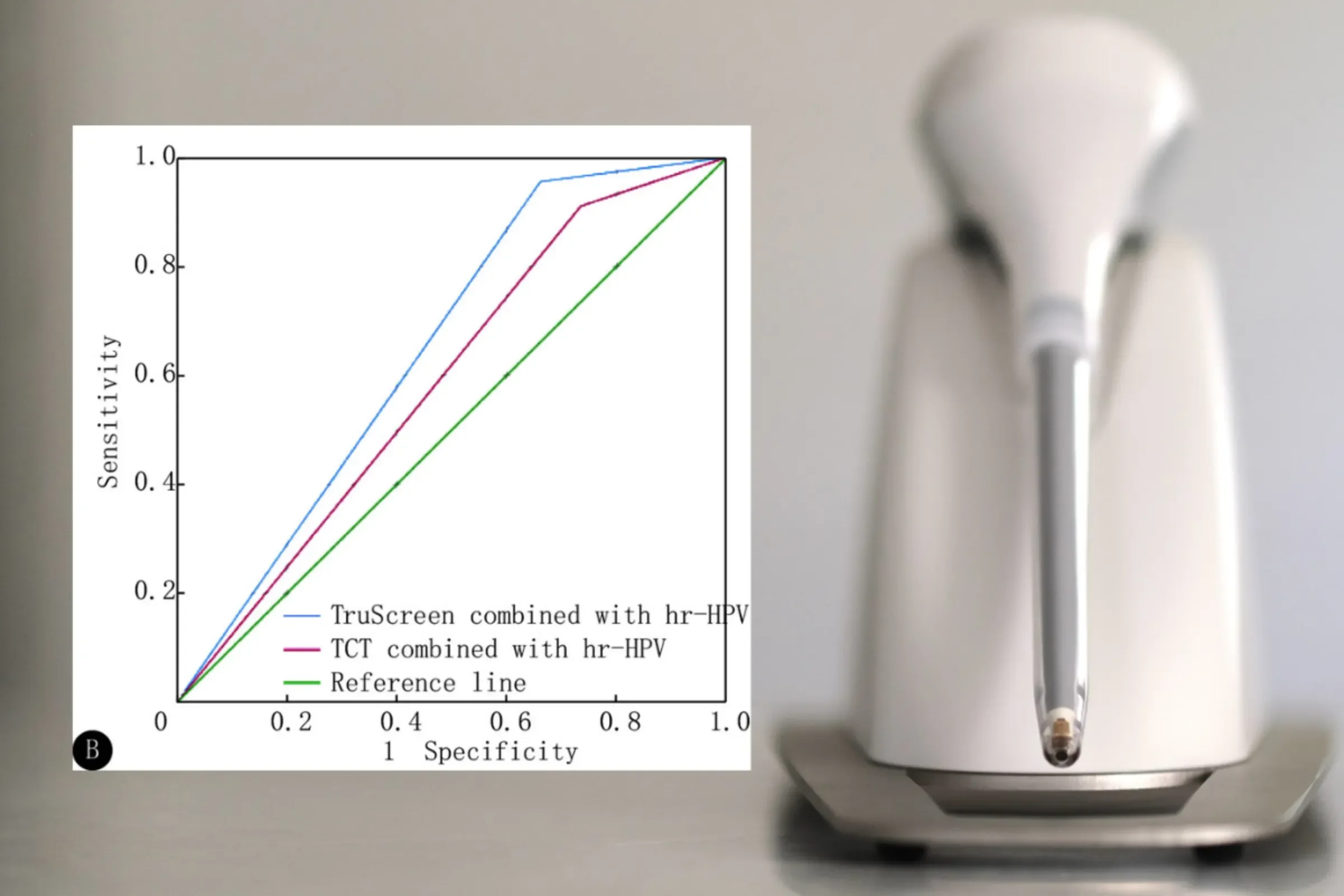
Sichuan University clinical study confirms superiorityof TruScreen + hr-HPV co-testing
An independent clinical study comparing TruScreen with Thinprep cytology test (TCT), and in combination with high-risk human papillomavirus (hr-HPV) testing, has been published and peer-reviewed in Journal of Sichuan University, confirming superiority of TruScreen + hr-HPV co-testing.
TruScreen validated as Superior Primary Screening Tool by World's largest Opto-Electronic Cervical Cancer Screening Study
Results of the world's largest ever (n=14,982) study of opto-electronic cervical cancer screening, conducted by Chinese Obstetricians and Gynaecologists Association (COGA), have been published validating TruScreen as a superior primary cervical cancer screening tool over Liquid Based Cytology (LBC) and high-risk Human Papillomavirus (hrHPV) testing.
Clinical study validates TruScreen’s efficacy and safety for pregnant women
Leading Chinese medical publication Family Doctor confirms TruScreen’s efficacy as a cervical cancer screening tool for pregnant women, in a 2,000-patient study conducted at Guilin People’s Hospital.
Published Saudi Arabia study confirmed TruScreen’s high sensitivity and specificity vs pap smear
Published Saudi Arabia study confirms TruScreen’s high sensitivity and specificity compared to pap smear.
Sign up for regular
updates and
announcements.
Can’t see the form?
Email us at marketing@truscreen.com to subscribe